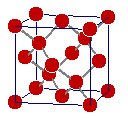
GRAPHITE |  DIAMOND |
Common crystal forms
- Cubic
- Orthorhombic
- Monoclinic
- Triclinic
- Trigonal
- Hexagonal
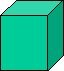
| Cubic - | contains three axes of equal length. All axes are positioned at right-angle to each other. A cube is also referred to as a closed form and can itself be a crystal. | |
|
| |
| Orthorhombic - | contains three axes which are of different lengths. All axes are positioned at right-angles to each other. | |

|
| |
| Monoclinic - | contains three axis of unequal length. Two axes are not at right angels. The third axis is at a right angle to the plane containing the other two axes and it is often referred to as the symmetry axis. | |

|
| |
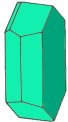
| Triclinic - | contains three axes. All axes are of different length and none at a right angle to the others | |
|
| |
| Trigonal - | contains four axes with three of equal length, all three arranged in the horizontal plane. The forth axis is perpendicular to that plane and is of a different length to the others. Axes and angles in this system are similar to the Hexagonal System. In the cross-section of a Trigonal crystal there will be three sides. | |

|
| |
| Hexagonal - | contains four crystallographic axes consisting of three equal horizontal, or equilateral axes at 120 degrees to each other, as well as one vertical axis which is perpendicular to the other three. This vertical axis can be longer or shorter than the horizontal axes. | |

|
| |